Diamonds are among nature’s most precious and beautiful creations.
ABOUT DIAMOND
Diamond forms under high temperature and pressure conditions that exist only about 100 miles beneath the earth’s surface. Diamond’s carbon atoms are bonded in essentially the same way in all directions. Another mineral, graphite, also contains only carbon, but its formation process and crystal structure are very different. Graphite is so soft that you can write with it, while diamond is so hard that you can only scratch it with another diamond.
OVERVIEW
58X HARDER
Diamonds are the hardest material on earth: 58 times harder than anything else in nature.
“LUCY” IN THE SKY
Fifty light years from earth, this star is a 10 billion-trillion-trillion carat diamond.
1 IN 1,000,000
The average yield in most diamond mines is 1 part diamond to 1 million parts host rock.
FACTS
- Mineral: Diamond
- Chemistry: C
- Color: Colorless
- Refractive Index: 2.42
- Birefringence: None
- Specific Gravity: 3.52 (+/-0.01)
- Mohs Hardness: 10
QUALITY FACTORS
The human contribution to a diamond’s beauty is a well-executed cut.
CLARITY
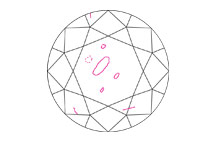
Clarity grades assess the number, size, relief, and position of inclusions and blemishes.
COLOR

The less color, the higher the grade. Even the slightest hint can make a dramatic difference in value.
CUT
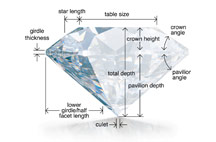
Cut (proportions, symmetry, and polish) is a measure of how a diamond’s facets interact with light.
CARAT WEIGHT

Rarity means larger diamonds of the same quality are worth more per carat.



